Learning Log
Chapter 9 stoichiometry
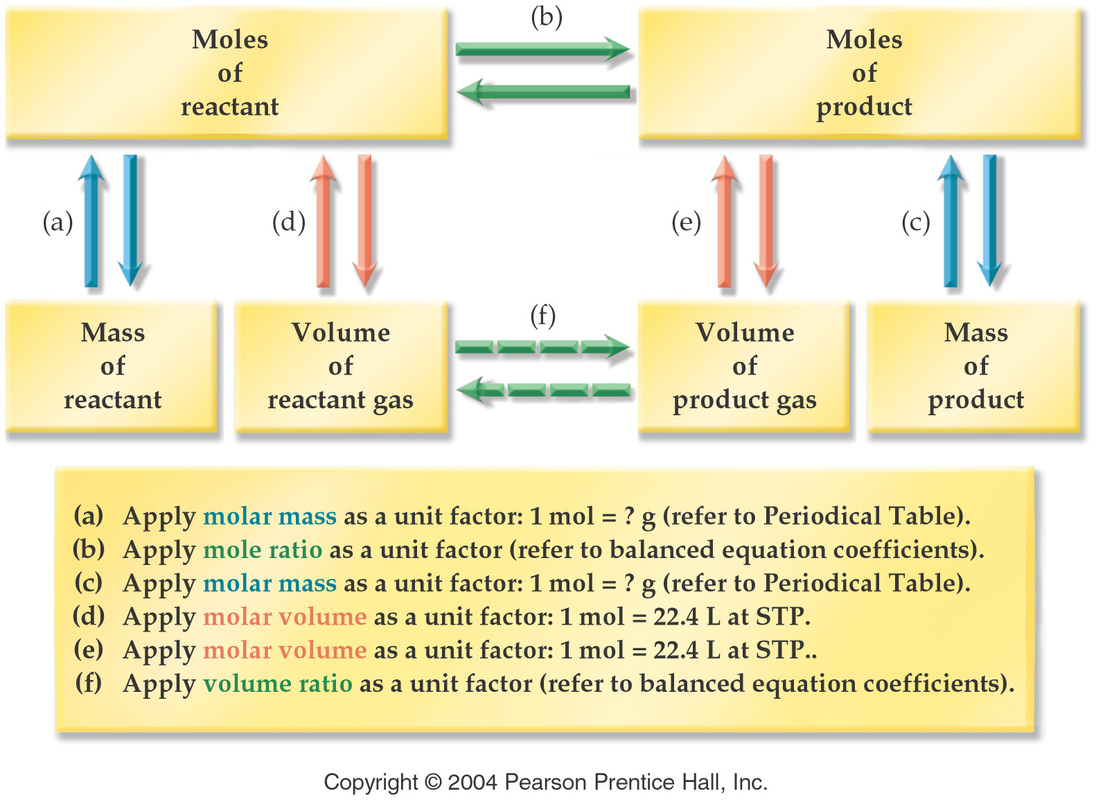
Stoichiometry is the name for calaculations that involve the relationships between reactants and products. To find these relationships scientists uses what is called a mole ratio. A mole ratio is the relatses the amount of two substances. The number one uses to create these ratios are the coefficents infront of each substance in an equation. To be able to use the ratio the most important aspect is that the equation must be balanced. Mole ratios are used to convert moles of one thing to moles of another. When one is given a problem when one sets up the mole ratio the unknown goes on the top and the given goes on the bottom. Also, in a an equation as a guide to wich way one would convert mole to moles, moles to grams, grams to moles or grams to grams would be a simple diaram such as GRAMS ->(divide by molar mass) MOLES->(mole ratio) MOLES->(multiply by molar mass) GRAMS. An example of an equation with all the steps would be: In the balanced equation 2Au_2O_3-> 4Au+ 3O_2, how many grams of gold can be made from the decomposition of 69.7 grams of gold (III) oxide? To solve this equation one first converts by dividing by the molar mass of Au_20_3 which is 441.94 like so 69.7g Au_2O_3/1 * 1mol/441.94g Au_2O_3. Next you multiply by the molar ratio -* 2mol Au/1mol Au_2O_3. Lastly you multiply by the molar mass by the seconds substance- * 196.97g Au/1mol. Then one sould do the math in your calculator and get the answer 62.1 grams of Au. Once you know how to convert using stoichiometry you can find the percent yeild. The percent yeild is the actual yeild divided by the theoretical yeild times one hundred. The threortical yeild is the calculations using the the conversions. The actual yeild is what one actually got when performing the experement. The percent yeild is ones accuracy to the perdicted yeild to ones own yeild with human error. An example of an percent yeild would be 40/48 * 100 would be a percent yeild of 83%. Remember when performing stoichiometry to check your converion factors by remebering to simplify.
Lab Abstract
This weeks lab was done to find the precent yeild of sodium chloride from the reaction of baking soda(NaHCO_3) and hydrochloric acid. In the lab the evaporating dish was weighed for it mass of 43.44 grams. Then the baking soda was measured for 0.50 grams and poured into the eveparating dish and the mass was taken for a third time of 43.93 grams (loosing .01 gams in the transpher process possibly?). Then the HCl was carefully and slowly dripped into the baking soda until the all baking soda was gone and the chemicals stopped reacting. Excess HCl may have been added due to inaccuracy. After the reaction the substances left are NaCl and H_2O. Then the evaporating dish was then placed over the bunsen bruned to boil off the water. MOre mass may have been lost during the boiling process from NaCl popping out of the dish. Little H_2O was left in the dish inexpess to prevent this from happening. However, the previous data became incurate when the evaporatind dish was remeasured to have a mass of 40.70. In an effort to correct the mistake the lone dish was reweighed but still had a mass of 41.68 grams. Due to the groups data being inacurrate the calculations we preformed with another groups data. The other groups data of 0.48 grams of NaHCO_3 was converted to 0.34 gams of NaCl using Stoichiometry to get the theoretical yeild.
The precent yeild was then found by dividing the other groups actual yeilds od 0.25 by the theoretical yeild to get a precent yeild of approximately 74%. If this lab was repreformed to be done better the balances should have been double checked and areas for loss of mass or added mass sould be avoilded with better care such as having percises amounts and times.
The precent yeild was then found by dividing the other groups actual yeilds od 0.25 by the theoretical yeild to get a precent yeild of approximately 74%. If this lab was repreformed to be done better the balances should have been double checked and areas for loss of mass or added mass sould be avoilded with better care such as having percises amounts and times.