learning log
chapter 5 periodic law
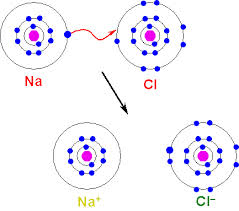
Chapter five started with the history of the periodic table. The first version of the periodic table was made by Dmitri Mendeleev in 1869 and was organized by atomic mass. However, there was a problem with this version. If you look closely on the periodic table some of the masses do not increase with each added proton and can vary. On the other hand Mendeleev also looked at the properties of each element and left some gaps where he predicted other elements or changed the order of the masses. It wasn't until 1911 that a scientist Henry Mosely reorganized the periodic table to what it is today after the newly discovered atomic number. The way the periodic table is set up there are clear trends fot the atomic radious, electronegativity, and ionization energy. The atomic radious gets smaller as one moves across left to right and larger as one moves down. This is because as it gose right there are more protons in the nucleus pulling the electrons closer to the nucleus and it gets larger as one moves down because every time a whole new energy level is added making the atomic radious bigger. All other trends are opposite the radious so electronegativity and ionic energy trends are greater across from left to right and smaller as one moves down. The reason elctrongativity is like this is because moving right the more attraction an atom has to other electons because the more desparate it is to finish its octet in its outer energy level. In the same way because as one goes down there are more energy levels creating more space between the electrons and protons thus having less attraction for electronegativity. For opposite reasons ionization energy has the same trend of being greater as one move to the right and less as you move down. While moving right an atoms attraction to its own increases to complete its octect and the more energy it takes to remove one of its electrons. Then as one moves down the less atraction it has to electrons and less energy it needs to take it. Once an atom has become an ion it throws off the balace and dramaticly changes the size of the atom. If it is a positive ion its called an cation and is half the size of its neutral counter part. This is because with more protons than electrons and the protons pull the electrons inward making the atom smaller. If an ion is negatlive it is double the size than if original and is called an anion. It is double the size because with more electrons than protons all the electrons repell eachother making the atom bigger.
Lab abstract
The periodic trends lab created a visual for the trends in the periodic table such as ionization energy, electronegativity, and atomic radious. Straws were cut to length baised off of data for each trend and then the data was analyzed. It was see that the radii decreased moving right across the periodic table because the more protons the more they attract the electrons inward making the radious smaller. Then the the radious increases as as you move down because each time a new energy level is added making it bigger. The elecrtonegativity increases as one moves aross to the right because the the closer it is to comleteing the octect and the more it wants more electrons to do this. As one moves down the periodic table the less electronegativity there is because the farther away the electrons the less atraction it has to its electrons and the less its atracted to more electrons. Lastly, ionization energy increases as it is moved across to the right because the more it wants to keep the electrons it already has making the ionization energy higher to take one away. It also decreases as one moves down the periodic table. This is because the farther away the electrons are making them less atracted and easer to yake away its electrons.(