LEARNING log
Chapter 4 electrons
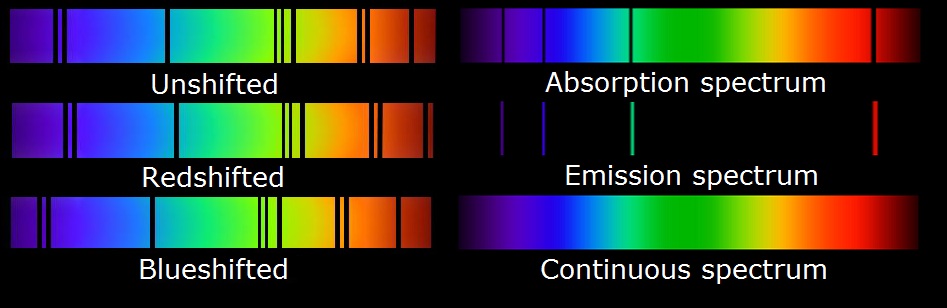
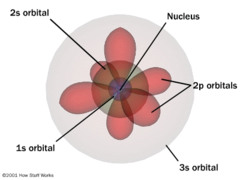
Chapter four is about electrons. First it is known that it is the energy in electrons and their energy that causes the light spectrum. There are many variations of light even kinds that one can't see. Light has the propreties of both waves and particles. Being described in waves, the more energy the light has the smaller the waves are and vice versa. In the areas of visable light violet waves are the shortest and red waves are the longest. However there are even stronger and weaker forms of light that we cant see. The higest energy wave are gama rays and the lowest energy waves are long waves. Electrons give off and take in energy by jumping to different energy levels. Moving from the old modle of the atom a new one was introduced that explains the transpher of atoms in the energy levels. It is known that it takes more energy for electrons to stay in the outer energy level and each level can hold a different number of electrons this is because of the orbitals in each level. There are seven total seven of all the atoms. The different orbitals in each level are called S, P, d, and f. Each of these have a different number of orentations that they can hold two electrons in. S has one oreintation, P has three, d has five, and f has seven. With each main level one orbital is added on (S,P,d,f) so an example would be 1S in level 1 or 2S 2P in level 2. We use quantum numbers to define electrons in an atom. There are four quantum numbers pricipal quantum number (the main energy level), angular momentum (the shape of the orbital), magnetic quantum number (the orientation of the orbital around the nucleus), and spin quantum number (the spin state of the electron). One can then use you knoledge of the energy levels to show certain elements such as carbon or iron. Due to each element having a unique number of protons and the same number of electrons by writing the orbitals in electron configurations such as 1S^2 2S^2 2P^4 (oxygen) and so on. Knowing which element it is one can just add up the exponents in the equation. ways to know when to stop when writing the configuration you can use orbital notation and noble gas notation.
Lab abstract
In the lab this week, the class analized the spectral lines of certain elements and compounds. It was started by annalizing sunlight and floresent light and contiued with hydrogen helium neon water and more. A sketch was made for each observation and data was recorded. The objective of this lab was to have a real life example of of the spectral lines and explanation of how spectral lines are created in each element and compounds.