learning log
chapter 18 chemical equilibrium
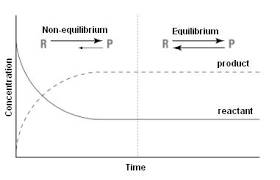
In a reaction there is a foward and reverce reactions where the reactants turn into products and products turn back into reactants. However, these foward and reverce reactions are not present in all reactions. An example when this cannot happen is when wood is burned, it does not have the product of smoke to create a reverce reaction floats up in the air. For both the foward and reverce reaction there is a rate at which it coures. If these reaction rates become equal and constant the reactions have achieved chemical equilibrium. Once these rates are the same the consentrations of both the reactants and products dont change, however, they do not have to be equal. If after equilibrium is achieved , but the reactions continue it is called dynamic equilibrium. The point at which chemical equilibrium is achived is repersented as K_eq. One can solve for K_eq in the equlibrium expression: K_eq= [C]^x*[D]^y/ [A]^n*[B]^m , where A and B are the reactants, C and D are the products, x and y are the coefficent of the products, and n and m are the coefficents of the reactants. An example of this equation is [1.1*10^-5]^2/ [6.4*10^-3]*[1.7*10^-3]= 1.1*10^-5. Certain factors can effect equilibrium by putting stress the reaction. These stresses can be changes in temperature concentration and if there are gasses pressure. Le Chatlier's Princpal helps explain these properties effect on reactions. It states: If a system at equilibrium is subjected to stress (change), the equilibrium shifs to relieve the stress. This means say the temperature in an endothermic reaction is increased for the products the equilibrium will shift to the the reactants or to the left of the reaction. Likewise if the temperature is increase to the rectants the equilibrium is shifted to right toward the products. If the temperature is decreased it will shift to the side on which the temperature is decreases to relieve the stress. If the consentration is added to either the reactants or products the reaction will shift opposite where the consentration was added. Chenging the pressure only effects gasses because their masses can be compressed and expanded. If an equation has both gasses and other states of matter only the gasses will decided what direction the reaction will shift. If the pressure is increased the reaction will shift to the side with less moles of gass, and vice versa for decreased pressure. One can tell the moles of a substance by looking at the coefficents. Lastly it is important to understand that a cattalist doesn't effect equilibrium it only increases the rate the equilibrium takes place.