Learning log
chapter 16 Therodinamics and chemistry
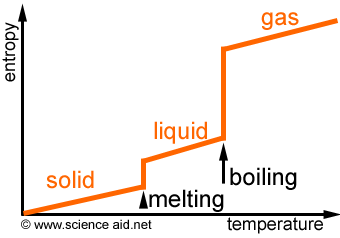
Thermochemistry is the strudy of the transphers of energy as heat that accompany chemical reactions and physical changes. A caloremeter is used to measure the energy absorbed or released heat. Temperature is the measure of the average kinetic energy of the particles in a substance. Kinetic energy is the energy possesed by an object due toits motion. Heat is the traspher of energy between objects because of a difference of temperature and is measured in joules as the SI unit. Heat is transphered from places of higher temperature to places of lower temperature. Specific heat is the amount of energy required to raise the temperature of one gram of a substance by one degree celcious or kelvin depending on which is given. The formula to find specific heat is Cp= q/m*delta T where q is the heat energy in joules delta is "chenge in" m is mass and T is temperature. One can also solve for the heat energy with the equation q=m* Cp* delta T. Delta T is calcultated by T_f- T_i= delta T which means the final temperature minus the initial temperature equals the change in temperature. An example of solving for Cp is Cp= 32J/4.0g*40k= 0.20J/g*k. An example of a q problem is q=10.0g*0.385J/g*k * 20= 77J. Reactions are either classified as exothermic or endothermic. Exothermic is when heat is given off and endothermic is when energy is absorbed. This energy is absorbed as heat. Enthalpy is the stability and heat given off when bonds are broken or formed. Endothermic reactions have high enthalpy while exothermic reactions have low enthalpy. Enthalpy writen in formula is delta H meaning change in heat. If delta H is shown negitive it is more stable and an exothermic reaction and vice vera if delta H is positive. Entropy is the measure of randomness of the particles, the disorder. Entropy will increases in a closed system is the second law of thermodinamics. The symbol for entropy is delta S. In a formula one can predict weither the products or reactants have the greater Entropy by looking at the number of products vs reactants and the states on either side the. The more substances on either side and the fastest moving matter (ie gas> liquid> solid) the more entropy ther is.
lab abstract
The lab this week was to calculate the specific heat of different metals and compare them. Each group had one metal and was compared to other groups metal. Iron was the the metal given and the mass was weighed. First the H_20 was heated to one hundred degrees celcious and the iron was placed inside. One the heat had come to an equalibrium. The metal was placed inside a double layerd styro foam cup and with water to cover the metal with the its temperature previously measured and mass weighed. The final temperature of the cup with the metal was recorded and the difference was delta T. The Cp of H_2O is 4.18. With this info the heat energy of the water was calculated which happens to also be the heat energy of the metal. Finaly the specific heat of the metal was calculated to appoximately 3.6. This was then compared to the actual specific heat of .449 and the percent accuracy was calculated. Of the metals Zinc, Iron , Aluminum, Copper, ect. aluminium was found to have the highest specific heat. This was then evaluated by the density and it was discovered that density and specific heat are indirectly related.