Learning Log
chapter 12 solutions
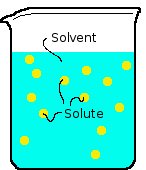
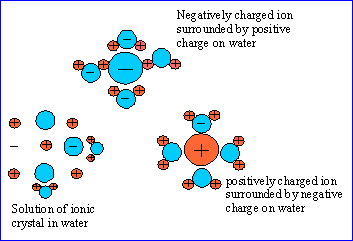
As stated in a prevous chapter there are two types of mixtures homogenous and herterogenous. Homogneous means the substances in the mixture are uniform throughout or equally mixed to the molecular level in all parts of the mixture. A heterogenous is the opposite where it is not mixed at the molecular level or is not is not mixed equally throught the whole mixture. An example of a homogenous mixture is sugar water while a heterogenous is milk or sand in water. A solution is a homogenous mixture of two or more substances in a single phase. In a soulution there is a solute and a solvent. The solute is a the substance being dissolved and has the lesser amount in the substance. The solvent is the substance that is the dissolving medium as well as the one with the greater amount. There are also types of mixtures called suspensions and colloids. A suspension is a mixture where the particles settle out whwen it's no longer stirred such as sand in water. A colloid is a mixture where particles are intermediate in size between those in solutions and suspensions. THe particles are larger than those in a solution but they stay mixed. Milk is an example of this because the fat molicule float evenly mixed in the milk but are not mixed at the molecular level this kind of colloid is called an emulsion. The actual definition of an Emusion is a fine dispersion of minute droplets of one liquid in another in which it is not soluble or miscible. When talking about solutes they can be classifined as electrolytes and nonelectrolytes. An electrolyte is a substance that dissolves in water to give a solution that conducts electricity. An example of this is salt. A nonelectrolyte is one that disolves in water to create a mixture that does not conduct electricity meaning it is a covalent bond. An example of this is sugar. There are different factors that can effect two substances rate of dissolution or how fast two substances form a solution. These factors include increasing the surface area of the solute, agitating the solution, and heating the solvent. When one increases the surface area of the solute it increases the the amount of area that the solvent then can break dissolve into itself at one time. When the solution is agitated or mixed (stirred, shaken, blended, etc.) it increases the rate of dispersion by dispersing the solute into the solvent making it easer to reach the surface area. When a the solvent is heated it causes the molecules to move faster so more can reach the surface of the solute. A solution has reached equlibrium it has reached the most possible amount of soulte the solvent can absorbe at one time due to the rate of the reaction equal to the rate of the opposing process tuning the solute back into its separated substance. When the substance has reached equlibrium it iss considered to be saturated and up until that point a soulution would be consided to be unsaurated. A soulutions solubility is its ability to become saturated under a certain tempature. The most common solvent is water. When somethind is dissolved in water or another substance the solute will only be dissolved if it has a like polarity. This means in water which is mixed with salt which is completely charged it will dissolve unlike oil which is nonpolar so it does not dissolve in water. This also works vice versa id the solute is nonpolar then oil would dissolve in it.
Lab abstract
The first lab that was preformed was the to see how the susrface area of the solute. This was preformed with NaCl in H_2O using five different types of NaCl varring in size. Each of these were stirred at an equal rate and timed for how long it took to disslove. The largest crystals too the longest taking over three minutes to to dissolve. The rest took under a minute and a half to full dissolve into the solution. The quickest to dissolve was a grinded normal salt futher grinded to a powder. When doing this experiment the procedure should have been followed more accurately for the group and a more stable constant stirring method.
The second lab that was preformed this week was solving for a consetration of H_2O to citric acid. Simple moles/liter conversion was used to find the amount of moles per liter for a certain amount of water. This was then converted to grams using the the valley of the moles. The citric acid was then measured using a ballance and poured into water and aggitated until dissolution was complete.
The second lab that was preformed this week was solving for a consetration of H_2O to citric acid. Simple moles/liter conversion was used to find the amount of moles per liter for a certain amount of water. This was then converted to grams using the the valley of the moles. The citric acid was then measured using a ballance and poured into water and aggitated until dissolution was complete.