Learning log
chapter 6 Chemical bonds
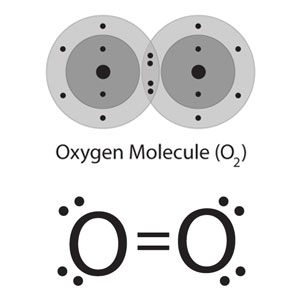
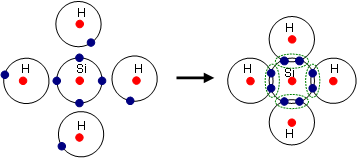
IN chapter six the focas of the week was chemical bonds. There are three different types of chemical bonds ionic, covalent, and metalic bonds. Each of these bonds combine with different attributes for each. Ionic bonds combine by giving or taking the valence electrons to or from the other atom. These bonds usually combine to form alternating positive and negative patterns to form what is called crystal latus structures. Once these combine they usually have high melting points, soulable in water, and are conductive in the soulution. An example of a ionic bond is NaCl or table salt. Another type of bond is covalent. These when elements combine they share electrons creating unique structres with mutiple elements. Properties of covalent bond include low melting points, soluable in water, and are not conductive in water or in a solid state. An example of a covalent bond would be water or sugar. Lastly, metalic bonds are when cations (obviously metals) are attached to each other with a pool or sea of electrons surrounding them. This type of bond makes a very high melting point, and are good conductors as a solid but not in a water solution. An example of a metallic bond is brass or steel. NInety nine precent of the time one can know what kind of bonds elements are making by looking at the periodic table. However it is also possible to know from the difference in electrongativity. If the difference is higher than 1.7 to 3.3 is ionic, 1.7 to 0.3 is polar covalent and 0.3 and below is nonpolar covalent. Nonpolar and polar tells wether some atoms in the bond are more attracted making some atoms more dominate than others making them slightly positive or negative repersented by delta +/-. Particualary covalent bonds can be represented by lewis dot structures. These are shown using the symbol and valence electrons in the shape of dots in pairs of two surrounding the symbol. One can then interpret the lewis dot structures into the the shapes of the actual atoms. The different structures include linear, trigonal planar, bent/angular, tetrahedral, trigonal pyramidal, ect.
lab abstract
The objective of the types of bonds lab was to find what kind of bond substances had baised on their properties. The substances used included salt, baking soda, sugar, sand, copper, aluminium, and oil. Baised on the information from the chapter it is known that ionic bonds have hight melting tempatures, souluble in water, and conductive in a water solution. Covalent bonds have low melting tempatures, souluable in water, but are not cunductive in water or in a solid state. Lastly metalic bonds have high melting tempatures, not soulable in water and is conductive as a solid. In the lab melting point, soulibility and condutivity. It was cconcluded that salt, baking soda, and an unknown substance all wereionic bonds, alumimium and copper were metalic bonds, and sugar, oil, and sand all had covalent bonds.