Learning log
Chapter seven using the chemical formula
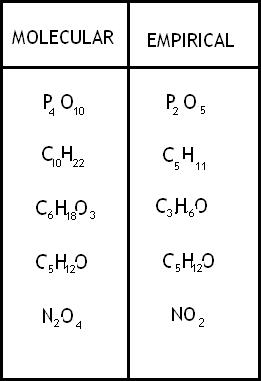
In chemistry the chemical formula is use to show which elements with however many atoms are combined to make a chemical. An example is H_2O where the subscribt is the number for atoms for H or hydrogen and one Oxygen atom. One can use the chemical formula to do conversions to moles and then to the number of molecules or ions. To do this one first needs to know the formula mass. To find the formuls mass simply add up all the atomic masses of each element for each atom. An example of this is CH_3COO witch would be (C) 12.01+(H) 1.01*3+(C) 12.01+16.00*2= 59.05amu. It is good to know that the formula mass is also the molar mass of the compound meaning 59.05g of CH_3COO equals one mole of the substance. Knowing this one can find how many moles of CH_3COO are in other amounts of grams. One can use a simple conversion factor to slove this. An example is take 30g of CH_3COO the conversion factor would be 30g/1 * 1mol/59.05g. One then solves this by canceling the grams and multiplying across to get 30mol/59.05= 0.508044mol and rounds off to 0.51mol. One can the take this a step further and find how many molecules make up 0.51mol. Another conversion is used like so 0.51mol/1 * 6.022*10^23molecules/1mol . This will then give one the number of 3.07122*10^23 and rounded off to 3.1*10^23molecules. Going back to the formula mass one can then find the precent composition of the element in the formula. This can be done by taking the amount of each element in the compond and dividing it by the formula mass. An example of this would be H_2O and the formula mass of water is 18.02g and 16.00g of it is oxygen and 2.02g of it is hydrogen. The first equation is set up like so 16.00/18.02=88.79% and the second equatio is set up like 2.02/18.02=11.21%. One can also do this the other way around solve for the formula mass with the precent composition. This is done by changind the precent to grams by assuming ther is 100g of the total formula (61%=61g) and dividing by the molar mass of the element to get moles and then dividing by the lesser element to get a ratio. This ratio then translates to the empirical formula or the smallest whole number ratio for a compound. An example to find the empirical formula is 31.9% K, 28.9%Cl, and 39.2% O. This then changes to 31.9g K 28.9g Cl and 39.2g O. Next you divide each by their molar mass 31.9g/1* 1mol/39.10g= .8158567775 mol K (don't round till the end), 28.9g/1 * 1mol/35.45g= .8152327221 mol Cl, 39.2g/1 * 1mol/16.00g= 2.45. One then divides the numbers to the tenths place by the smallest number to get .8152327221= 1 Cl, .8158567775=1 K, and 2.45=3. The ratio is then 1:1:3 and its empirical formula written as KClO_3. However, if the ratios were not perfect whole number such as 1:1:2.5 one would multiply (In this case by 2) to get a whole number ratio such as 2:2:5 to get a formula of K_2Cl_2O_5.
lab abstract
The lab this week burnt a piece of magnesium to create magnesium oxide. This was preformed by using the bunsen bruners to dry the crucibles and then let cool before putting the magnesium in the crucibel over the burner. Before the magnesium and crucibel was put over heat the mass was taken on the balance. After one minute on the bunsen burner water was added to stop the magnesium reacting with nitrigen and only with oxygen. Ater another thirty seconds on the burner the crucible with magnesium was left to cool and then meaured for mass again. Contrary what was believed would happen the total mass actually increasedue to the added mass of the oxygen. The mass was then converted to moles and then solved for the empirical formula. The accepted formula for magnesium oxide was MgO wich five out of the eight groups got for high accuracy. This experiment tied into chapter seven by finding the formula fusing only the mass of the substance.