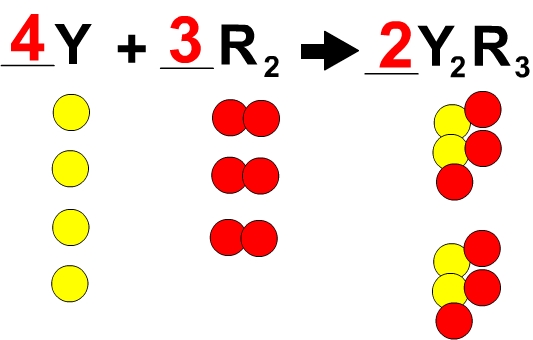Learning log
Chapter 8 chemical equations
The week of chemical equations started with uderstanding the vocabulary. In a chemical equation there are the reactants and the products. The equation is set up like so: reactants-> products. The reactants are the substances before the reaction, the arrow means yeilds or becomes, and the products are the substances after the reaction. An example of this would be H_2+ O_2 -> H_2O_2. Ther are then added symbols to signify what state the reactants and products are these include (s) solid, (l) liquid, (g) gas, or (aq) mixed with water. There are then added symbols to the yeilds sight to tell what kind of energy was added to cause the reaction. Some include a triangle for heat, an two arrows pinting foward and back maning it is reversable, or hv over the yealds sign naming light is added. There are four indications that a chemical reaction has happened evolution of energy (heat or light), production of gas, formation of a percipitate, and/or a color change. Also, when one writes a chemical equation one needs to follow the law of conservation of mass. The law of conservation of mass states that atoms are neiither created nor destroyed during an ordinary chemical reaction. One can follow this law by balancing an equation. This means you add coefficents which add another whole molecule to either the reactants or the products to make them equivalent. When you do this if a single atom has a subscript you also need to multiply the subscript by the coefficent. An example of balancing an equation is 2H_2+ 2O_2-> 2H_2O + O_2 when before it was like H_2+O_2-> H_2O+ O_2 which was unbalanced. There are four different kinds of chenical reactions synthesis, decomposition, single displacement, double displacement, and combustion. A simplifed example of these would be A+X-> AX (synthesis), AX-> A+X (decomposition), A+BX-> AX+B (single displacement), AX+BY-> AY+BX (double displacement), and Fuel+ O_2-> CO_2+ H_2O (combustion). A synthesis is when two substances combine to create a single composition and a decomposition is the opposite when one composition reacts and creates two seprate substances. Single displacement is when one substance replaces another in a composition during a reaction. A double displacement is when who compouunds have one substance that switch places with each other creating two different compounds. Lastly a combustion is when a fuel like propane or wood reacts with the oxygen in the air giving off carbondioxide and water into the air.
Lab abstract
In the lab this week nine different substances were mixed with one another to see if a reaction occured if any to overall find what the unknown substance was. This was done by comparing the reactants to the unknow if they had a similar type of reaction. The reactives were nitric acid, silver nitrate, sodium iodide, calcium carbonate, sodium hydroide, Iron (III) nitrate, copper (II) nitrate, and acidic acid. After preforming the the tests between the eight substaces the unknown was then tested against all other eight substances. Once the substances were all tested a comparison was made between how the substance reacted with the unknown and with eachother. The closest to the reaction of the unknown was NaOH reacting with Fe(NO_3)_3 creating an opeak white and then to yellow a yellow percipitate with fizz and AgNO_3 creating an orngish brown percipitate. This was just like the unknown and because of which the group came to the conclusion that the unknown was NaOH.