Learning Log
chapters 14 and 15 acids and bases
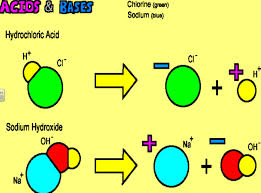
Acids and Bases are substances that either have H+ (acids) or OH- (bases) when dissolved in H_2O. Properties of acids include a sour taste, creating hydrogen gas with active metals, turn blue litmus paper red, strong acids combine with strong bases to make water and a salt, and have a charge. Bases properties include being slippery, changing red litmus paper blue, a stong base mixed with a strong acid make water and a salt, has a charge, and tastes bitter. pH and pOH are used to measure the strength of H and OH atoms in a solution. On the pH scale and vise versa for the pOH scale the acides are between 0-6 and bases are 8-14 on the scale. Equilibrium is the is when the dissolution and opposite rection have come to a point where they are happening at an equal rate (Further explained in Learning Log 12). One can use the constant of H+ an OH- equilibrium to fint the Molarity of H+ or OH- atoms. This constant is show by K and is the number 1.0*10^-14. When one is given a the molarity of either H+ or OH- such as 1.0*10^-6 is M of H+ the formula to use is K=[H+][OH-]. If one inserts the numbers 1.0*10^-14= 1.0*10^-6 [OH-] just divide the value of H+ from K to get 1.0*10^-8. One can also simply find the pH or pOH of an caid or base from the molarity. The formula for pH or pOH is pH(/pOH)= -log [M or pH or pOH]. The seven strongest acids are hydriodic acid, perchloric acid, hydrobromic acid, hydrochloric acid, sulfuric acid, chloric acid, and nitric acid. When these strong acids mix with strong bases such as LiOH, NaOH, or KOH a salt and H_2O is formed. A salt is an ionic compound of a cation and an anion bonded by opposite charge. This is why salts and H_2O are often neutral.
lab abstract
The objective of the lab was to determine the substane with the highest, lowest, and near neutral pH. To accomplish this 10mL of each ten substances were poured into a beaker and 5mL of cabbage juice was used to predict weather the pH from color. Then the pH meter was used to accurately find the pH. The substance with the highest pH was 11.03 meaning it was a base. The substance with the lowest pH was Muratic Acid with 1.25 meaning it was an acid. These two strong acid and base were then mixed together to to neutralize the substance back to blue purple color.